Like Bristol-Myers Squibb, AstraZeneca has maintained a “pure-play” pharma approach to business in recent years as well as a preference for smaller, targeted “bolt-on” acquisitions, rather than the large-scale M&A pursued in recent years by Pfizer, Merck and Roche.
Some Wall Street analysts suggest that may be about to change, as the U.K. multinational announced Aug. 28 that Pascal Soriot, a chief architect of Roche’s successful absorption of Genentech, will take over as its new CEO on Oct. 1, replacing acting CEO Simon Lowth and succeeding David Brennan, who stepped down in April.
With AstraZeneca struggling through a severe patent cliff – Seroquel (quetiapine) lost U.S. patent protection earlier this year, with Nexium (esomeprazole) slated to follow in 2014 and Crestor (rosuvastatin) in 2016 – full-year worldwide pharmaceutical sales are projected to decline by 16% this year. Bernstein Research analyst Tim Anderson wrote following the Soriot announcement that AstraZeneca’s five-year financial outlook is “uninspiring,” due to a thin late-stage pipeline and a mixed track record of R&D success.
“Doing small bolt-ons, like AstraZeneca has previously described, probably won’t be enough for the company to change its trajectory quick[ly] enough,” Anderson opined in an Aug. 29 note. “Doing a larger acquisition (>$20B in size) would more immediately accomplish this goal, in our view, and AstraZeneca would likely be able to borrow an amount of this size such that its dividend would remain secure and its share buybacks might even continue.”
Bolt-on transactions have been the order of the day recently at AstraZeneca. The firm has been active in acquiring companies with late-stage or marketed products to bolster its flagging R&D pipeline, the most recent being the joint acquisition with Bristol of Amylin Pharmaceuticals. With the purchase, AstraZeneca, which will incur a cost of around $3.5 billion, and Bristol obtained two marketed anti-diabetic GLP-1 agonists, Byetta (exenatide) and Bydureon (exenatide extended-release).
AstraZeneca also announced the $1.27 billion acquisition of Ardea Biosciences to gain access to the Phase III gout drug lesinurad, on June 23. Along with bolt-ons, the company’s business strategy has focused heavily on returning cash to shareholders through share buybacks. Brennan, who announced he was stepping down on April 26, after the company reported an 11% decline in revenues year-on-year in the first quarter of 2012, was an advocate of the buybacks strategy.
But Anderson believes Soriot will be emboldened by his experience in merging Roche with Genentech, a process in which insiders say the exec successfully managed both the process and the people. “He knows both the science and the commercial side of the pharmaceutical business, and he proved himself to be a leader when folding in Genentech,” Anderson wrote. “The Roche/Genentech tie-up continues to function well, all things considered. With this experience in hand, could he be tempted to do a bigger deal at his new company?”
Another analyst, Eric Le Berrigaud of Bryan Garnier & Co., does not see major philosophical changes in the near term, however. “Soriot comes from a company that drives a pure-play strategy in the pharmaceutical industry, which suggests a change in strategy from this perspective at AstraZeneca is unlikely, i.e., no diversification on the horizon.” Just the same, Le Berrigaud also predicts that smaller-scale business development activity will continue at AstraZeneca at its recent hectic pace.
So there you have it – either Pascal Soriot will be a change agent at AstraZeneca in favor of big M&A to turn around the pharma’s prospects … or he won’t.
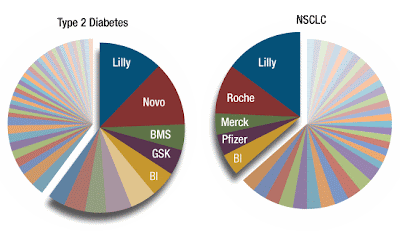
Yet another perspective, from our sister publication PharmaAsia News, posits that Soriot will try to replicate at his new address the strong emerging markets performance enjoyed by Roche. Particularly in China, Roche has soared in emerging market sales growth, while AstraZeneca has underperformed the industry, and in a business segment it identifies as a priority, no less,
Roche has shunned branded generics in its emerging market strategy – a business pathway pursued vigorously by AstraZeneca – and instead focused on selling innovative specialty products such as high-priced biologics, despite the potential reimbursement hurdles. The strategy has paid off – Roche reported 37% year-over-year sales growth in China during the second quarter, while AstraZeneca sales grew by 12% in that country and only 1% worldwide on the quarter.
As always, your crack deal-watching team has labored to bring you the latest edition of:
Sunovion/Elevation – Sunovion Pharmaceuticals will pay $100 million upfront and up to $430 million total to buy out privately held Elevation Pharmaceuticals in a deal announced Aug. 30. Central to the deal is Elevation’s Phase IIb candidate for chronic obstructive pulmonary disease (COPD), EP-101, which combines an aerosolized formulation of glycopyrrolate with a proprietary inhaler device, the eFlow Nebulizer System. In addition to the upfront, Sunovion (the CNS/respiratory disease-focused company created by the merger of U.S. specialty pharma Sepracor and Japan’s Dainippon Sumitomo) will pay Elevation up to $90 million in development milestones, up to $210 million in commercial milestones and potentially another $30 million if other Elevation programs are developed successfully. Elevation claims that EP-101 is the only nebulized, long-acting muscarinic antagonist (LAMA) bronchodilator in development – the intended indication is for moderate to severe cases of COPD. Phase III trials are slated to begin in the second half of 2013. The buyout provides an exit for Elevation’s venture capital backers, including Canaan Partners, Novo Ventures, TPG Biotech, Care Capital and Mesa Verde Venture Partners. Founded in 2008, Elevation raised roughly $44 million over two financing rounds. Most recently, the biotech announced a $30 million Series B in early January, but had only drawn down a $12.4 million first tranche of that round at the time of the sale’s announcement. – Joseph Haas
Janssen/Genmab – Janssen Biotech has turned again to Genmab to boost its R&D pipeline, this time licensing worldwide development and commercialization rights to the Danish biotech's clinical-stage, CD38-targeted monoclonal antibody daratumumab, in a deal valued at a hefty $1.1 billion. For that sort of money, one would expect a lot of competition for rights to the molecule, and indeed that was the case – Genmab's CEO Jan van de Winkel claimed to be still receiving approaches from other companies the day before he sealed the deal with Janssen on Aug. 30. More than a dozen companies apparently expressed an interest in the asset. The agreement includes an upfront license fee of $55 million, an equity investment of $80 million and up to $1 billion in development, regulatory, and sales milestones, in addition to tiered double-digit royalties. Just weeks before, the two companies had concluded a much smaller deal, valued at $175 million, under which Janssen Biotech will evaluate Genmab's newer bispecific antibodies against a range of therapeutically relevant targets. Daratumumab targets CD38, found widely on the surface of multiple myeloma cells, and it has the potential to be a first-in-class drug for the treatment of multiple myeloma and other hematologic cancers. Initial data have shown it offers a potent and broad spectrum of anti-cancer activity. Janssen looks to be the ideal partner, as it already markets a multiple myeloma therapy, Velcade (bortezomib), in some countries. Genmab has executed a noted turnaround in fortune, after needing to slim down and restructure in 2009-2010. Now, all three of its active clinical-stage products have big pharma partners, and its cash runway extends out four years, at which point royalty and licensing revenues just might be large enough to sustain the company. – John Davis
Alnylam/Monsanto – Alnylam Pharmaceuticals has inked an agreement with big agriculture firm Monsanto that puts Alnylam’s intellectual property in the service of developing next-wave pesticides and other agricultural products. The deal announced Aug. 28 brings Alnylam a $29.2 million upfront payment with the potential for downstream milestone payments and royalties, along with research funding. In exchange, the companies have an exclusive 10-year arrangement in which Monsanto will apply Alnylam’s RNA interference technologies to its BioDirect platform, which aims to bring innovative biological solutions to farmers. Alnylam in turn sent $1.4 million of the upfront to Isis Pharmaceuticals, under a 2004 arrangement in which Alnylam licensed IP for double-stranded oligonucleotide therapeutics that mediate RNAi. Isis also is in line to receive a portion of any milestones or royalties that Alnylam earns under its partnership with Monsanto. According to Alnylam Chief Business Officer Laurence Reid, the agreement with Monsanto is ideal because it gives his company a new source of non-dilutive funding from which it can finance its internal RNAi therapeutic development program, dubbed “5x15.” Alnylam staff will be involved somewhat in technology transfer efforts with Monsanto, but the agriculture firm will do most of the work and will provide research funding for whatever assistance it needs from Alnylam. Neither company would provide much in the way of specifics about how RNAi would be used to create next-wave agricultural products, with a Monsanto spokesperson saying it was still “early days” for determining what kinds of products might evolve from the partnership. “Our collaboration with Alnylam relates to work we have in discovery,” she said. Such products might be used to control devastating agricultural pests and diseases. Monsanto has calculated the annual worldwide market for agricultural biologics at about $1.7 billion. – JAH
Hospira/Orchid – For the second time, injectable generics seller Hospira struck a deal with India’s Orchid Chemicals & Pharmaceuticals to boost its antibiotic-manufacturing abilities. Hospira said Aug. 29 that it would pay about $200 million to acquire Orchid’s 12-year-old, 50,000-square-meter active pharmaceutical ingredient manufacturing facility in Aurangabad, India, which houses 640 chemists, engineers and technologists. The deal also includes a research and development facility in Chennai, home to 160 scientific professionals. Hospira says the acquisition will support manufacturing of key antibiotic products, including wide-spectrum beta-lactam antibiotics such as imipenem-cilastatin and injectable meropenem. The new deal is expected to close in the fourth quarter, and be break-even to slightly accretive to earnings in the first year following its close. In a deal that closed in March 2010, Hospira paid $381 million for another of Orchid’s antibiotic-manufacturing complexes and associated R&D facilities, where it manufactures finished-dosage-form injectable generics. Orchid plans to keep its cephalosporin manufacturing business, which supplies Hospira with that drug. – Paul Bonanos
Merz/Shionogi – Merz Inc., the U.S. affiliate of Merz Pharmaceuticals GMBH, announced Aug. 27 the acquisition of Cuvposa (glycopyrrolate, coincidentally the same active ingredient in Elevation’s COPD drug, EP-101, noted above) from Shionogi & Co. Financial terms were not disclosed. Cuvposa is an oral solution for sialorrhea, over-excretion of saliva, specifically for pediatric chronic severe drooling associated with neurological conditions such as cerebral palsy. Cuvposa indirectly reduces the rate of salivation by preventing stimulation of acetylcholine receptors located on peripheral tissues, including salivary glands. FDA approved Cuvposa as an orphan drug in 2010, and it has been available in the U.S. through specialty pharmacy since April 2011. It is the only FDA-approved treatment for this condition. Merz, Inc. CEO Bill Humphries said the acquisition of Cuvposa “is a promising addition to our neurology business and reflects our commitment to becoming a recognized leader in the treatment of movement disorders and related conditions.” Merz, Inc. develops and commercializes products in the U.S. for its German parent, and its portfolio comprises neurology, aesthetics and dermatology products. – Michael Goodman
Photo credit: Roche